AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Gamp category 1 examples8/31/2023   
As a result, GAMP covers all aspects of production from the raw materials, facility and equipment to the training and hygiene of staff. 
One of the core principles of GAMP is that quality cannot be tested into a batch of product but must be built into each stage of the manufacturing process. More specifically, the ISPE's guide The Good Automated Manufacturing Practice (GAMP) Guide for Validation of Automated Systems in Pharmaceutical Manufacture describes a set of principles and procedures that help ensure that pharmaceutical products have the required quality. This can be documented within the validation plan or the risk assessments.Good automated manufacturing practice ( GAMP) is both a technical subcommittee of the International Society for Pharmaceutical Engineering (ISPE) and a set of guidelines for manufacturers and users of automated systems in the pharmaceutical industry. For example a Laboratory Instrument (Category 3 – COTS) which is pre-use and post-use calibrated or runs standard along with the test need less verification than a system where only the results are relied on. While GAMP5 provides guidance to the approach based on the categories there are better rationales that can be put in place rather than the complexity of the software. This implies a level of governance to be applied over suppliers independent of the maturity or complexity of the software. “Vendor documentation, including test documents may be used as part of the verification documentation, providing the regulated company has assessed the vendor” GAMP5 still includes these categories however the benefits are not integrated within a Science and Risk Based Approach to validation and the ASTM approach. 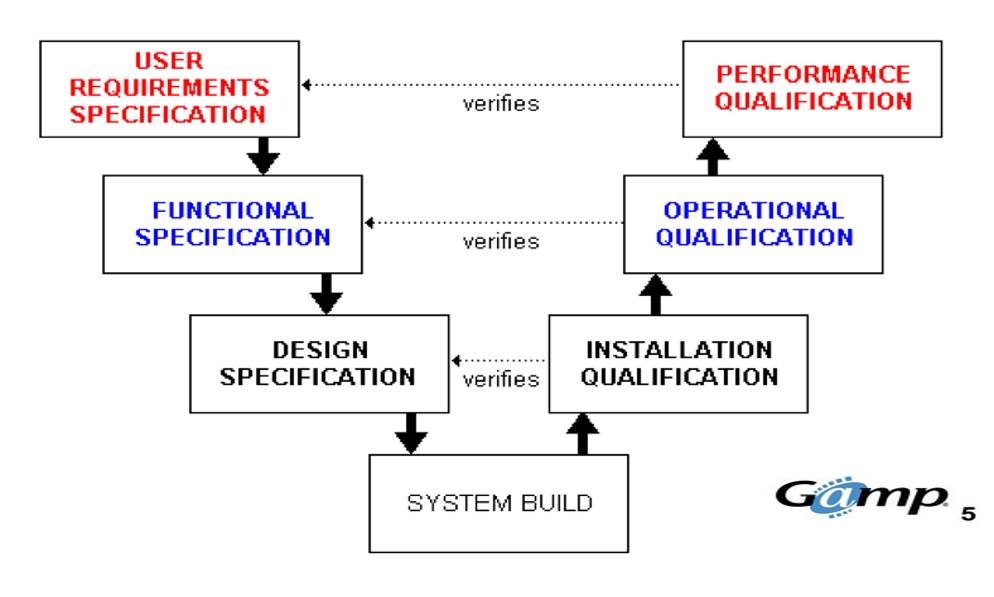
Categories 1-3 were considered to standard systems and the System Life Cycle Design (SLCD) documentation were not required, this included In GAMP3 and 4 the purpose of the GAMP categories had clear purpose, identifying which validation deliverables were not required. However as technology has advanced the it has been recognised that complex software can be embedded (firmware) within systems. At the time that GAMP4 was issued firmware was considered to be used for simple instruments. These have been revised in GAMP5 to four categories as detailed below:Ĭategory 1 – Infrastructure software including operating systems, Database Managers, etc.Ĭategory 3 – Non configurable software including, commercial off the shelf software (COTS), Laboratory Instruments / Software.Ĭategory 4 – Configured software including, LIMS, SCADA, DCS, CDS, etc.Ĭategory 2 from GAMP4 has been removed. In GAMP4 there were five software categories. The GAMP categories were originally introduced to provide an initial assessment as to the validation requirements / deliverables. The complexity and the maturity of the software can be used to support and mitigate identified risk but should not be used to determine the validation / verification deliverables. The software categories identified in GAMP5 do not fit with determining the risk to product quality, efficacy or data integrity and no longer plays an integral part to determining that a computer system is fit for purpose. As discussed in ISPE GAMP5 the GAMP Categories for hardware and software have been retained in GAMP5, all be it in a modified format from GAMP4. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
